Researchers Progress in Developing Vaccine for Pathogenic Staphylococcus aureus
August 29, 2025 feature
by Delthia Ricks, Phys.org
contributing writer
edited by Gaby Clark, reviewed by Robert Egan
scientific editor
associate editor
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Antibiotics are the old medicine cabinet standby for treating infections caused by multidrug-resistant Staphylococcus aureus, but as antimicrobial resistance continues to mount globally, scientists say there's a need for new strategies.
While vaccines are a potential answer, achieving an effective way to immunize against multidrug-resistant S. aureus has led scientists down dozens of blind alleys. Ten candidate vaccines that looked promising in preclinical animal studies in recent years failed miserably in human clinical trials.
Now, scientists in China are investigating a way to sidestep the myriad problems that plagued vaccine investigators in the past by choosing not to target a whole antigen. Instead, they say, it's time to home in on a critical 'surface loop' as a vaccine target. The infinitesimal loop is located on the S. aureus antigen known as MntC.
The loop is an ideal target, scientists in China say, because it has been shown to be essential for aiding survival in S. aureus by mitigating oxidative stress. By triggering vaccine-induced antibodies that zero in on that site, survival is impossible. As a vaccine target, the surface loop is known as an epitope.
Writing in Science Translational Medicine, researchers at institutions from throughout China report on the creation of a first-of-its-kind vaccine that has been tested in animal models based on a target pinpointed in samples from human clinical trials. The evolving strategy overcomes past obstacles and confers protection against drug-resistant S. aureus with a vaccine.
Past attempts to develop a vaccine against S. aureus have failed because S. aureus is among commensal microbial communities that thrive in the human microbiome. S. aureus is a common colonizer of the skin, the nasal passages, and less commonly, the gut. The presence of S. aureus as a commensal bacterium renders it a 'nonprotective immune imprint' in humans.
A non-protective immune imprint refers to a phenomenon where a person's immune system develops a memory of harmless commensal bacteria. That memory can hinder effective immune responses against the more dangerous pathogenic strains of the bacteria. MRSA (methicillin-resistant Staphylococcus aureus), for example, is a drug-resistant form of S. aureus.
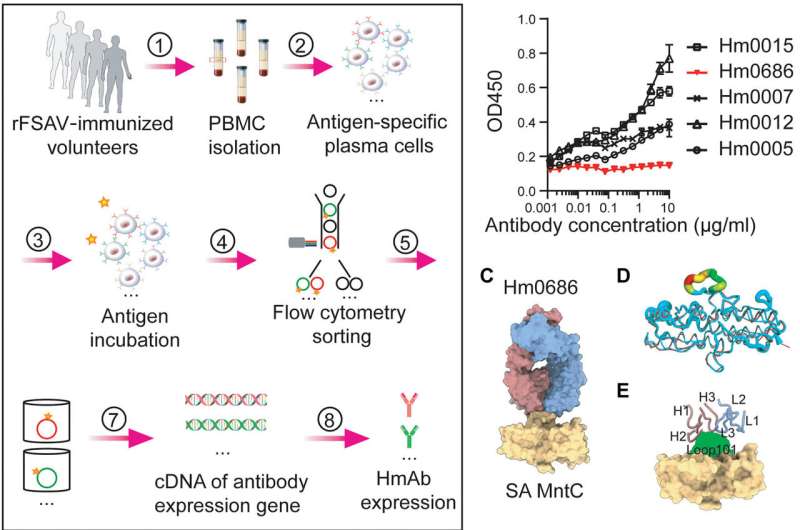
'Humans, as natural carriers of Staphylococcus aureus, have developed nonprotective immune imprints that can be reactivated by S. aureus antigen vaccination and that contributes to the failure of S. aureus vaccine trials,' writes Xiaokai Zhang, lead author of the new research, explaining why previous candidate vaccines did not work.
Zhang is an immunologist in the Department of Microbiology and Biochemical Pharmacy at the College of Pharmacy and Laboratory Medicine of the Third Military Medical University in Chongqing, China.
S. aureus is common in human populations around the globe, and an estimated 70% to 80% of people worldwide are believed to harbor non-protective immune imprints against S. aureus, Zhang and colleagues report.
With such a widespread presence of nonprotective immune imprints, there was little doubt, according to the team, why previous vaccine attempts failed.
'To test whether an epitope-focused vaccine strategy can overcome this issue, we explored the protective epitope of the notable S. aureus antigen MntC,' Zhang continued. 'A surface loop of MntC, Loop101, was found to be essential for S. aureus to absorb manganese ion and survive oxidative stress.'
By using MntC's Loop101 as a target, the team in China found a way to exploit one of the bacterium's survival mechanisms as a vaccine target. The team also studied similar vulnerabilities in other forms of bacteria, such as Escherichia coli and Streptococcus pneumoniae.
In S. aureus, MntC is a surface-bound lipoprotein that functions as the ligand-binding component of the MntC manganese transporter. This protein is crucial for the bacterium's survival by enabling it to capture manganese, which is also critical for resisting wayward assaults by the host's immune system.
Zhang and colleagues found that their epitope-based vaccine induces high titers of anti-Loop101 antibodies. In the animal model portion of their investigation, the team also found that the epitope vaccine protects mice that were pre-exposed to S. aureus.
'On the basis of our findings, several important implications for the development of Staph aureus vaccines can be drawn,' Zhang wrote in conclusion. 'First, the components of Staph aureus vaccine candidates are often relatively conserved. Identifying functional regions of the antigens that play essential roles in the pathogenicity of S. aureus could substantially accelerate the subsequent screening process for protective epitopes.'
'Second, the study suggests that the evaluation of vaccine efficacy should not be limited to testing the concentration of binding antibodies against the whole antigen. Instead, it should focus on examining antibodies specific to the 'critical functional site,' such as Loop101, which provides a more precise measure of the vaccine's immunoprotective effect.'
Written for you by our author Delthia Ricks, edited by Gaby Clark, and fact-checked and reviewed by Robert Egan—this article is the result of careful human work. We rely on readers like you to keep independent science journalism alive. If this reporting matters to you, please consider a donation (especially monthly). You'll get an ad-free account as a thank-you.
More information: Xiaokai Zhang et al, An epitope vaccine derived by analyzing clinical trial samples safeguards hosts with prior exposure to S. aureus against reinfection, Science Translational Medicine (2025). DOI: 10.1126/scitranslmed.adr7464
© 2025 Science X Network




